Process Analytics in the Biotechnology and Pharmaceutical Industries
Biopharmaceutical manufacturing is characterized by the use of modern technologies and scientific progress, as well as highly complex research and development work
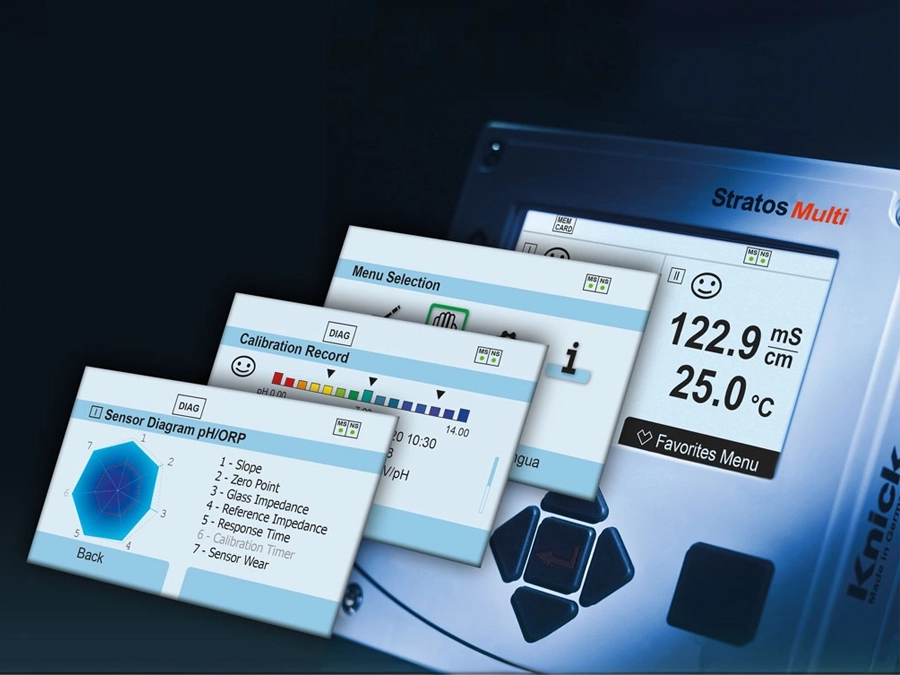
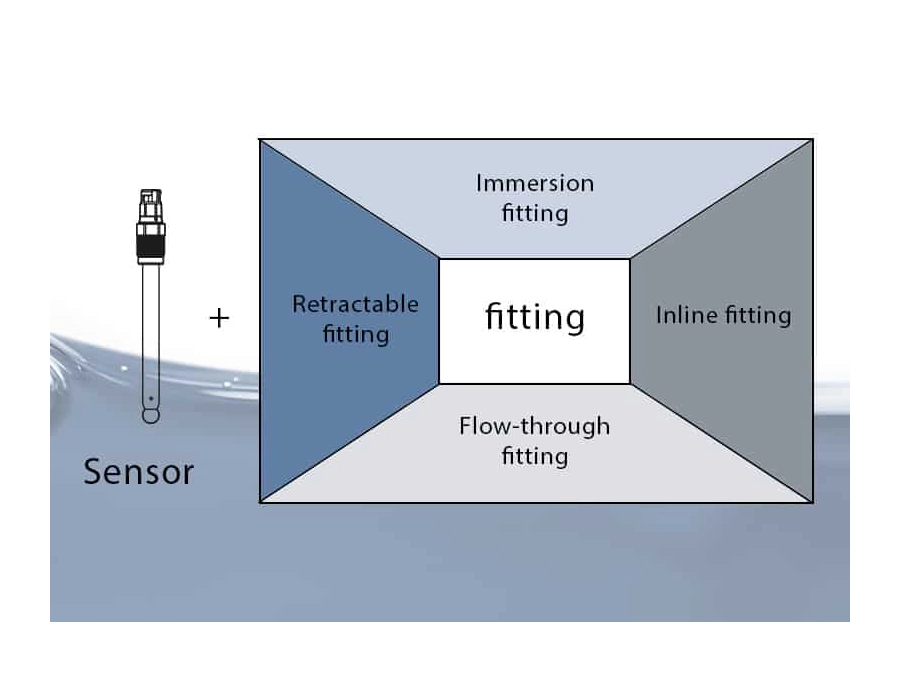
For decades, Knick has consulted and supported companies in process analytics worldwide. The company’s key competencies include measuring the pH value, ORP, conductivity, and dissolved oxygen – with process reliability. Alongside sensors, Knick supplies innovative industrial transmitters, high-quality process connections and fittings, and unique cleaning and calibration systems.

Progressive Technologies Driven by Highly Complex Research and Development
As a rule, developing an innovative active pharmaceutical ingredient requires a huge amount of time and capital in order to convert scientific discoveries into new medication and set up specialized production systems with the right equipment. Initial investigational medicinal products must be manufactured for clinical trials and then the ingredient must receive FDA approval before full production can begin.
Today it is virtually impossible to treat rare or complex diseases without treatments developed by the biopharmaceutical industry. Animal and insect cells, bacteria, and yeast are genetically modified in order to produce target molecules like recombinant proteins, vaccines, and much more. Due to the heterogeneity of the living organisms, production in this field is a complex process. The heterogeneity of the upstream process is also transferred downstream to the subsequent product cleaning steps.
The automation of all upstream, downstream, and supporting processes contributes to reducing the risk of human error, increasing operational efficiency, and in turn, increasing the safety and quality of the end product.
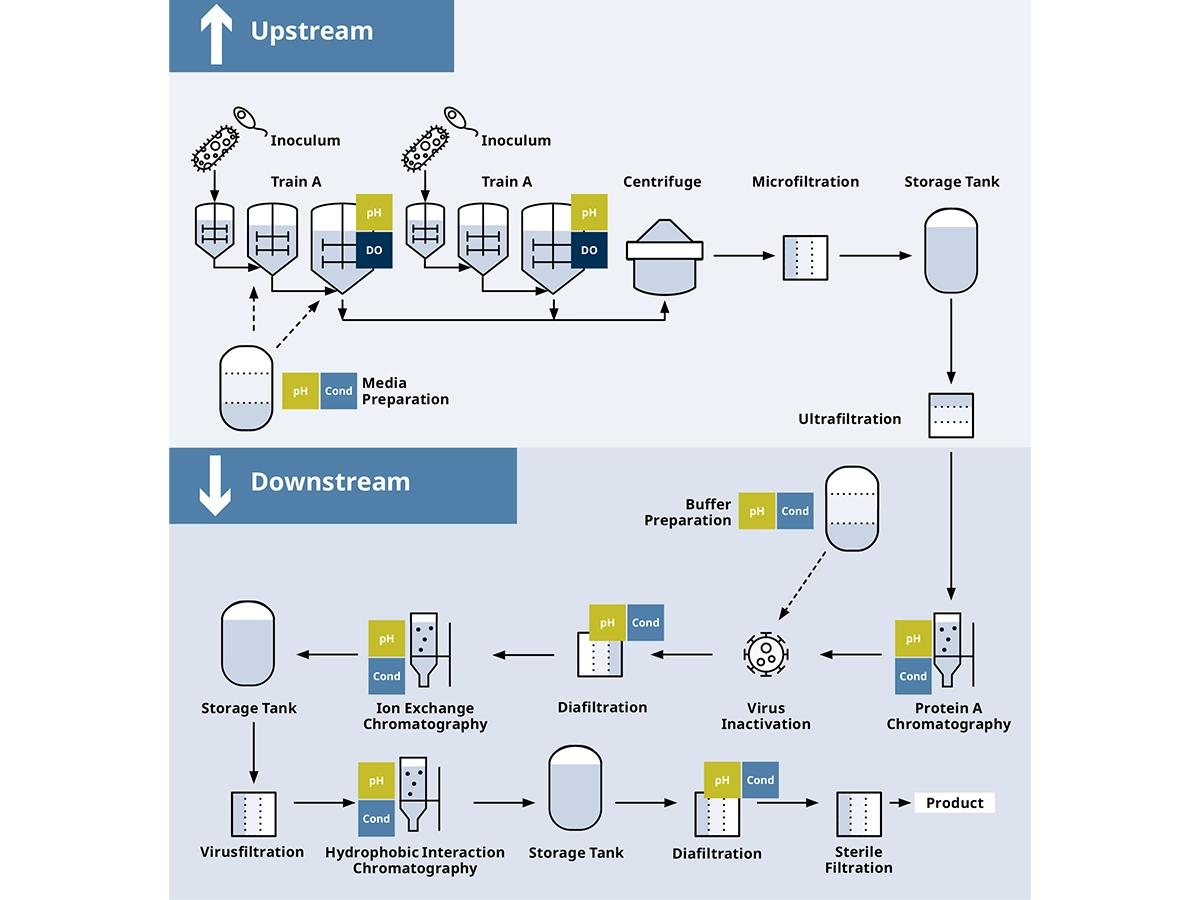
Downstream
Water for Injection WFI
ChemPharma
Upstream
Why Knick?
As a specialist for measuring and control technology, Knick offers a high level of competency and application know-how in the area of liquid analysis in demanding applications. Knick’s production depth ensures product quality and flexibility when it comes to customer-specific requirements.
A customer focus and reliability characterize the company, as do innovative products and technologies that are used worldwide by renowned manufacturers in the chemical and pharmaceutical industries.
Biopharmaceutical Process: Solutions for Every Process Step
In the biopharmaceutical industry, every process step is important and requires its own measuring technology in order to control critical parameters for maximum yield and as few deviations as possible.
Knick offers complete measuring loops from sensors to fully automated sensor maintenance systems. Automated processes contribute to raising standardization, avoiding transcription errors, and improving compliance.
With the cCare system from Knick, key goals of the FDA’s PAT initiative can be met:
- Standardization of calibration and cleaning processes to reduce fluctuations and deviations
- Elimination of transcript errors and therefore, fewer defective batches
- Thanks to Audit Trail, compliance with the applicable regulations

Biopharmaceutical Industry Brochure
Overview of specific applications and product solutions in various areas of the
Related Applications
Do you have more questions? We are here for you.
Contact us!